The market for hydrogen is vast consisting of recent demand for renewable energies and industrial and chemical processes needs such as methanol production, ammonia (NH3), removing sulfur from gasoline, float glass production, edible fat hydrogenation, weather balloons, rockets, etc. Today hydrogen is predominately generated as a by-product of the release of CO2 in the processing of fossil fuels.
Water electrolysis can be accomplished by solar driven electric voltage across a semiconducting p/n junction. However, in water, hydrogen is strongly bonded to oxygen and a minimum voltage of 1.229 eV (at 25°C, 1 bar) is required for splitting water into hydrogen and oxygen. This energy is larger than the bandgap of many semiconductors. For example, the badgap of silicon is 1.1 eV, therefore, a single silicon p/n junction cannot produce sufficient energy for breaking the O-H bond.
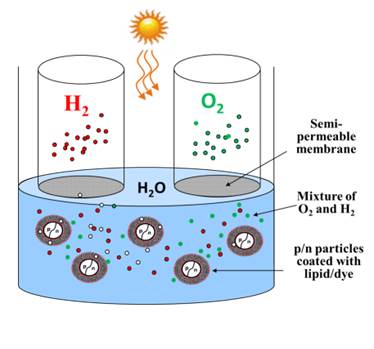
Figure 1: Schematic diagram of the proposed solar hydrogen generator.
We have recently discovered that silicon p/n particles coated with a functionalized biomaterial membrane can electrolyze water when dispersed in water and exposed to light. The membrane is made of lipid, oxidized cholesterol, and Merocyanine dye. The membrane catalyzes the electrolysis by providing a proton motive force (PMF) that adds to the solar driven electron motive force (EMF) of the p/n particles. Solar driven action of the proposed electrolyzer, simplicity of the system, large surface area of nanoparticles each acting as a nano-electrolyzer, and the lack of the costly electrodes as shown in Figure 1 are the main advantages of the proposed approach.
Representative Publications
- Energy harvesting capability of lipid-merocyanine macromolecules: A new design and performance model development, L. Tayebi, M. Mozafari, R. El-khouri, P. Rouhani, D. Vashaee, Photochemistry and Photobiology, DOI: 10.1111/php.12193, 2013
- Photothermal Hydrogen Production with Hybrid Organic/Inorganic p-n particles, D. Vashaee, L. Tayebi, A Parikh (patent pending)
Achievements
To support our claim and verify the ability of nanoparticles to split water and generate hydrogen, we built a prototype system. We used silicon as a starting material. Although silicon is not the best material for our purpose, due to its low voltage and oxidation issue, it is a reasonable and readily accessible test material to test (1) if lipid/MC coating can provide the additional voltage required to split water, and (2) if a multijunction p/n Si bead can also be used to split water molecules without the lipid/MC coating. For this purpose, we made two solutions of water and the Si beads.
(1) if lipid/MC coating can provide the additional voltage required to split water, and (2) if a multijunction p/n Si bead can also be used to split water molecules without the lipid/MC coating. For this purpose, we made two solutions of water and the Si beads.
In one solution, the beads were left uncoated and in the other they were coated with lipid/MC layer. Interestingly, we observed bubble formation in both solutions, which indicates water electrolysis for both systems. However, the bubble formation was significantly enhanced in the second solution which contained lipid/MC coated beads (Figure 2).
The fact that uncoated beads can also split water is consistent with the notion that nanoparticles are sticking together, which in turn makes multiple junctions of p/n that would enhance the total voltage above the threshold value of 1.229V required to split water. Moreover, the role of lipid/MC coating in further catalyzing the process is also confirmed in this experiment.
We have filed a patent application for this invention. The research was funded by OSU TBDP program and we have two pending proposals on this topic.

Figure 2: Bright field image of the bubbles formed around the p/n nanoparticles from water splitting. In (a) the Si beads are uncoated and in (b) they are coated with lipid/MC dye. The significant enhancement in bubble formation is seen in (b) which confirms the catalyzing role of lipid/MC coating.

